
How to Manage Coffee oxidation for Maximum Shelf Life Success
Coffee powder stales faster than beans because grinding increases the surface area by over 1,000%, allowing oxygen to attack flavor-carrying lipids and volatiles instantly. You open a fresh bag expecting a rich, complex aroma, but instead, you are met with a flat, cardboard-like disappointment that ruins your morning ritual. Understanding the chemical mechanics of coffee oxidation is the only way for roasters and consumers to ensure their specialty brew remains as vibrant as the day it was roasted.
How does surface area impact coffee oxidation?

Grinding breaks the bean’s protective structure into thousands of microscopic particles, exponentially increasing the exposure points for coffee oxidation. A whole bean acts like a natural vault, shielding its internal oils from the surrounding atmosphere. Once that vault is shattered, oxygen reaches every fraction of the coffee mass simultaneously.
The kicker is:
- Whole beans have a low surface-to-volume ratio, slowing gas exchange.
- Grounds expose the delicate internal matrix to air in seconds.
- Fine powder creates a massive “attack surface” for reactive oxygen species.
Why do smaller particles stale faster?
The smaller the particle, the less distance oxygen has to travel to reach the core.
Why does this matter?
- Diffusion happens almost instantly in fine grounds compared to whole beans.
- Moisture in the air hitches a ride on the increased surface area.
- Smaller fragments cannot hold onto the protective CO2 layer required for freshness.
Summary Table:
| Feature | Whole Beans | Coffee Powder |
|---|---|---|
| Surface Area | Low / Compact | High / Exponential |
| Oxygen Exposure | Surface only | Total mass exposure |
| Freshness Window | 2–4 Weeks | 15–30 Minutes |
This comparison highlights why pre-ground coffee loses its soul significantly faster than whole beans.
Key Takeaway: Grinding coffee removes the physical barrier to air, making immediate brewing or advanced packaging essential for flavor preservation.
Why do aromatics vanish during coffee oxidation?
Aromatics vanish because the volatile organic compounds (VOCs) that create scent are released the moment the bean’s cellular walls are crushed during coffee oxidation. These gases are highly unstable and evaporate into the air rather than staying trapped within the coffee structure. Without a protective environment, the “freshly roasted” smell is lost to the room.
It gets better:
- VOCs provide the nuanced floral and fruity notes in high-end coffee.
- Oxidation converts these fragrant molecules into odorless or foul-smelling compounds.
- Grinding creates heat, which further excites these molecules and speeds their exit.
Can we trap aromatics after grinding?
While you cannot stop evaporation entirely, you can slow the rate of loss significantly.
The reality is:
- Cooling beans before grinding helps retain a fraction more of the volatile oils.
- Immediate vacuum sealing can “freeze” the aromatic profile temporarily.
- Most ground coffee loses 60% of its aromatic intensity within 15 minutes of exposure.
Summary Table:
| Component | Freshly Ground | Oxidized Powder |
|---|---|---|
| Aromatic Volatiles | 100% | < 20% (after 1 hour) |
| Flavor Profile | Complex / Vibrant | Flat / Muted |
| Perceived Quality | Specialty Grade | Commercial / Stale |
The rapid loss of VOCs is the primary reason why ground coffee smells better than it tastes if left out.
Key Takeaway: Aromatics are the most fragile part of the coffee experience; once they evaporate, no brewing method can recover them.
Does CO2 loss accelerate coffee oxidation rates?
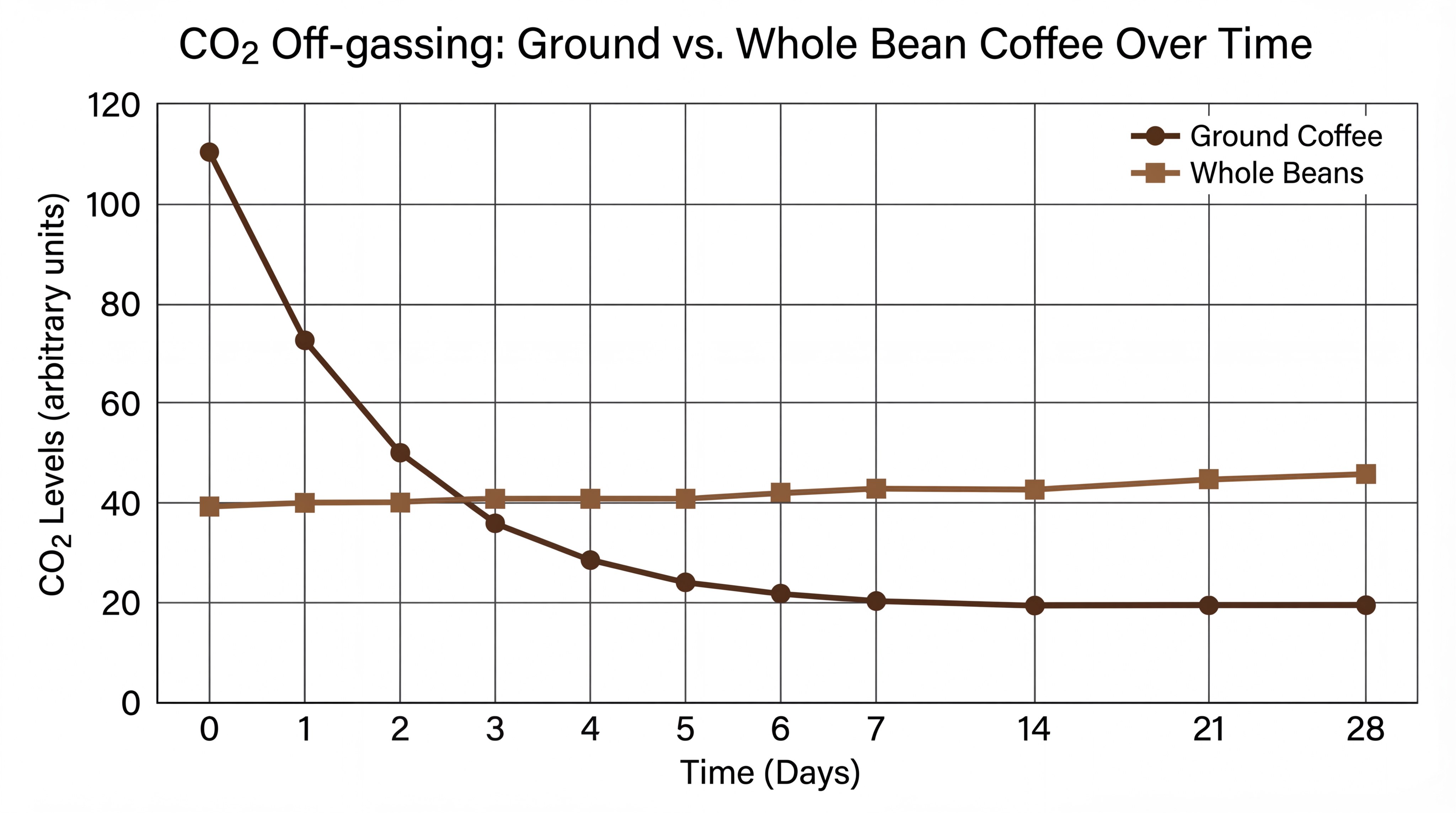
Yes, because carbon dioxide (CO2) acts as an invisible shield that physically pushes oxygen away from the coffee’s surface, and losing it facilitates coffee oxidation. During roasting, beans build up internal gas pressure that slowly leaks out over weeks in whole beans. Grinding releases roughly 80% of this protective gas within a single minute.
Think about this:
- CO2 creates a “buffer zone” inside the packaging.
- The lack of CO2 allows oxygen molecules to penetrate deep into the coffee’s pores.
- Degassing is necessary for brewing, but it is the precursor to staling.
Why is the “bloom” important for freshness?
The bloom is the visual evidence of CO2 being displaced by hot water during brewing.
Here is the deal:
- A vigorous bloom indicates the coffee still has its gas shield intact.
- Ground coffee that doesn’t bubble is already heavily oxidized.
- The absence of gas means the flavor-protecting barrier is completely gone.
Summary Table:
| Gas Status | Impact on Flavor | Protection Level |
|---|---|---|
| High CO2 (Fresh) | Bright Acidity | High (Blocks Oxygen) |
| Low CO2 (Stale) | Bitter / Flat | None (Open to Air) |
As the chart illustrates, the drop-off in CO2 for ground coffee is nearly vertical compared to beans.
Key Takeaway: Carbon dioxide is your coffee’s natural preservative; once it’s gone, the clock on oxidation accelerates.
Can lipid breakdown trigger coffee oxidation?

Lipid breakdown triggers a cascade of staling because the fats in coffee turn rancid when they are exposed to oxygen during coffee oxidation. These oils are responsible for the body and mouthfeel of your cup. In whole beans, lipids stay tucked inside the cellular matrix, but grinding smears them across the surface of every particle.
You see:
- Oxidized lipids create that specific “old peanut” or metallic taste.
- Darker roasts have more surface oils, making them even more vulnerable.
- Lipid oxidation is an exothermic reaction, meaning it generates its own heat to keep going.
Does roast level change oxidation speed?
The darker you roast, the more porous the bean becomes and the more oil is pushed to the surface.
But wait, there’s more:
- Light roasts have a tighter structure that resists oxygen longer.
- Dark roasts are essentially “pre-opened” for oxidation due to high roasting heat.
- Oily beans should never be ground until the very second they are needed.
Summary Table:
| Roast Level | Lipid Exposure | Rancidity Risk |
|---|---|---|
| Light | Internal / Low | Moderate |
| Medium | Mixed / Surface | High |
| Dark | Visible Oil | Severe |
The visible oils on dark roasted beans are like magnets for atmospheric oxygen and moisture.
Key Takeaway: Your coffee’s fats are what make it taste rich, but they are also the primary source of foul off-flavors.
How does humidity speed up coffee oxidation?

Humidity speeds up the process because water molecules act as a catalyst that helps oxygen molecules penetrate the coffee particles during coffee oxidation. Coffee is hygroscopic, meaning it actively pulls moisture out of the air. This moisture dissolves the coffee’s flavor compounds and begins the chemical breakdown before you ever start your brewer.
The bottom line is:
- High humidity opens the pores of the coffee grounds further.
- Moisture allows for secondary chemical reactions like hydrolysis.
- Damp grounds lose their aromatic intensity in a fraction of the time.
Is moisture the silent flavor killer?
Most people worry about air, but moisture in a humid kitchen can be just as damaging.
Believe it or not:
- Coffee stored near a sink or stove is at constant risk.
- Condensation inside a bag can trigger mold growth in ground coffee.
- Even a 1% increase in moisture content can halve the shelf life.
Summary Table:
| Environment | Moisture Level | Oxidation Rate |
|---|---|---|
| Arid / Dry | < 10% | Slow / Controlled |
| Moderate | 40-50% | Standard |
| Tropical / Humid | > 70% | Near-Instant |
Moisture and oxygen work together to strip coffee of its sweetness and replace it with sourness.
Key Takeaway: Controlling the humidity in your storage area is just as vital as excluding air to prevent staling.
Will heat exposure worsen coffee oxidation?

Heat exposure worsens the situation because it provides the kinetic energy needed for oxygen molecules to react faster with coffee compounds during coffee oxidation. According to the Arrhenius equation, the rate of chemical reactions generally doubles for every 10°C increase in temperature. This means a bag of ground coffee on a hot counter stales twice as fast as one in a cool pantry.
The truth is:
- Heat causes the gases inside the coffee to expand and escape faster.
- High temperatures accelerate the rancidity of natural coffee lipids.
- Sunlight adds UV radiation, which breaks down molecular bonds in the grounds.
Should you store coffee in the fridge?
Storing coffee in the fridge is often a mistake due to temperature fluctuations.
Listen to this:
- Every time you open the bag, moisture from the fridge air condenses on the cold coffee.
- This moisture then catalyzes oxidation as soon as you put it back.
- A steady, cool room temperature is superior to a fluctuating cold one.
Summary Table:
| Storage Temp | Effect on Grounds | Resulting Flavor |
|---|---|---|
| 15°C (Cool) | Stable Molecules | Fresh / Bright |
| 25°C (Room) | Moderate Reaction | Standard Aging |
| 35°C (Hot) | Rapid Breakdown | Bitter / Dull |
Thermal energy is the engine that drives the degradation of every cup you brew.
Key Takeaway: Heat is a force multiplier for oxidation; keep your coffee in a dark, cool spot to buy yourself time.
Why is bean porosity key to coffee oxidation?

Bean porosity is key because the honeycomb-like structure of roasted coffee provides millions of microscopic hiding spots for flavor, which coffee oxidation ruthlessly targets. Roasting transforms the dense green bean into a brittle, porous sponge. When you grind the coffee, you expose these “inner chambers” that were once inaccessible to the air.
Why does this matter?
- The internal surface area of a bean is vastly larger than its external skin.
- Porosity increases the deeper the roast goes.
- Grinding turns a three-dimensional protective structure into a two-dimensional exposed map.
Does the grind size affect internal access?
The finer the grind, the more you shred the internal pores of the bean.
The fact is:
- Coarse grinds (French Press) retain some of the original “honeycomb” structure.
- Fine grinds (Espresso) essentially destroy every internal wall.
- Finer grounds provide oxygen with an “all-access pass” to the coffee’s core.
Summary Table:
| Structure Type | Particle Size | Air Penetration |
|---|---|---|
| Intact Pore | Whole Bean | Surface Only |
| Cracked Pore | Coarse Grind | Moderate |
| Shattered Pore | Fine Grind | Total |
The microscopic architecture of the bean is the final defense against environmental decay.
Key Takeaway: Grinding is the destruction of the bean’s physical defense system, leaving no part of the coffee safe.
Can packaging prevent rapid coffee oxidation?

Advanced packaging is the only effective way to stop coffee oxidation in pre-ground products by creating a modified atmosphere that excludes oxygen entirely. At NewIdea Pack, we focus on engineering barriers that manage the internal gas environment. High-quality packaging uses nitrogen flushing and one-way valves to ensure the coffee remains in a “stasis” until the consumer breaks the seal.
Here is how it works:
- One-way valves allow CO2 to escape without letting oxygen in.
- Multi-layer laminates block 100% of UV light and moisture.
- Nitrogen flushing replaces active oxygen with an inert, non-reactive gas.
What should you look for in a bag?
Not all coffee bags are created equal when it comes to longevity.
Check this out:
- Look for a thick foil lining that prevents light transmission.
- Ensure the bag has a functional degassing valve near the top.
- Resealable zippers are essential for keeping air out after the first opening.
Summary Table:
| Package Feature | Primary Function | Freshness Impact |
|---|---|---|
| Degassing Valve | Releases gas pressure | Essential |
| Nitrogen Flush | Removes active O2 | High |
| UV Barrier | Blocks light decay | Moderate |
Sophisticated packaging technology can extend ground coffee’s life from minutes to many months.
Key Takeaway: The bag is more than a container; it is a high-tech climate-control system for your flavor.
Does grind size influence coffee oxidation?

Yes, the finer the grind size, the faster the coffee oxidation because fine particles provide the maximum possible surface area for chemical reactions. As shown in the image, the visual quality of the crema—which relies on trapped oils and gases—is the first thing to degrade when using oxidized grounds. Fine espresso grinds can become noticeably stale in as little as 15 minutes if left exposed to open air.
The kicker is:
- Coarse grinds have a slower staling rate because more flavor is “locked” inside the chunks.
- Fine powder has zero internal protection remaining.
- The extraction quality of espresso depends entirely on the presence of unoxidized lipids.
Why is espresso so sensitive?
Espresso requires high pressure to emulsify oils, and oxidized oils simply will not emulsify properly.
Why does this matter?
- Stale grounds produce “crema” that is thin, watery, and disappears quickly.
- Oxidation creates bitter, ashy notes that are amplified by high-pressure brewing.
- Freshly ground coffee is non-negotiable for high-quality espresso extraction.
Summary Table:
| Grind Type | Surface Area | Staling Speed |
|---|---|---|
| Coarse (Cold Brew) | Lowest | 1–2 Days |
| Medium (Drip) | Moderate | 2–4 Hours |
| Fine (Espresso) | Highest | 15–30 Minutes |
Managing the time between grinding and brewing is the single most important factor for home baristas.
Key Takeaway: The finer you grind, the faster the clock ticks; never grind more than you are about to brew.
Is shelf life tied directly to coffee oxidation?

Shelf life is a direct measurement of how much coffee oxidation has occurred and how many original aromatic compounds remain in the bean. While coffee never “spoils” in a way that makes you sick, its peak culinary window is incredibly narrow. Whole beans can maintain their profile for 4 weeks in a simple bag, but ground coffee hits its shelf-life limit in days.
It gets better:
- “Best by” dates on supermarket bags often refer to a year, but flavor is long gone.
- Real shelf life is about the ratio of volatiles to oxidized lipids.
- Even in a sealed bag, a tiny amount of residual oxygen will slowly degrade the coffee.
How do you know if your coffee is dead?
You don’t need a lab to tell if your coffee has lost its life.
Just look for these:
- The aroma smells like old nuts, wood, or nothing at all.
- The coffee tastes purely bitter with no acidity or sweetness.
- There is no bubbling or “bloom” when you add hot water.
Summary Table:
| Coffee State | Peak Flavor Window | “Dead” By |
|---|---|---|
| Whole Bean (Sealed) | 4–8 Weeks | 6–12 Months |
| Whole Bean (Open) | 2 Weeks | 1 Month |
| Ground (Open) | 15 Minutes | 2 Days |
True freshness is a fleeting window that requires active management of the storage environment.
Key Takeaway: Fresh coffee is a perishable agricultural product; treating it like a dry shelf-stable good is a recipe for bad flavor.
*
Conclusion
The science reveals that the shift from whole bean to powder is a race against the atmosphere. We have solved the mystery of why flavor vanishes so quickly by looking at surface area, gas loss, and lipid decay. By understanding these principles, you can take control of your coffee’s quality and avoid the “cardboard” taste of stale grounds.
NewIdea Pack’s vision is to push the boundaries of preservation technology so that every roaster can deliver perfection to their customers’ doors.
FAQ
Can I stop oxidation by putting coffee in the freezer?
No, you can only slow it down, and only if the bag is vacuum-sealed to prevent moisture from condensing on the grounds.
What’s the best container for ground coffee?
The best container is an opaque, airtight canister with a one-way valve or a vacuum seal, kept far away from any heat sources.
How do I know if my coffee has oxidized?
Directly smell the bag; if the vibrant, fruity, or chocolatey notes are replaced by a scent of old paper or wet wood, it is oxidized.
Is pre-ground coffee always stale?
Yes, unless it was packed immediately using professional nitrogen-flushing technology and remains sealed in its original bag.
Can I “save” oxidized coffee?
No, once the chemical bonds of the oils and aromatics are broken, they cannot be restored, though cold brewing can sometimes mask the stale notes better than hot brewing.
If you need help optimizing your packaging line to fight off the effects of oxygen
Please contact us today. We are dedicated to ensuring that the science of oxidation never stands between you and the perfect cup of coffee.
